Her name was Amina

They say you never forget the first patient you lose. Mine was an 11 months old little girl, during my first rotation in pediatrics as a house officer. Her parents brought her to the pediatrics emergency department in Yaoundé – Cameroon, unresponsive with a high fever. She had been ill for two days and had failed to improve with several home remedies tried by her parents. I held her pale body in my arms as the skilled pediatrics nurse and pediatrics attending searched her limp arms and legs for venous access. The diagnosis – severe Malaria complicated by severe anemia. The time it took to get venous access and start an emergent blood transfusion probably wasn’t that long but, in the moment, felt like a thousand hours.
Despite the medical team successfully starting the blood transfusion and IV treatment with antimalaria medications, a few hours later Amina will go into cardiac arrest and lose her life. This moment has remained burned into my memory in vivid detail. Maybe it is the heart-breaking sobs of her mother begging us to save her child, or the defeated helpless figure of her quiet father in the back of the room or the anguish in Amina’s eyes as life slipped away from her. Maybe it is all of these elements together and my own helplessness in the moment which make this memory so powerful – I remember her name, her face, and all the emotions like it happened yesterday. I also remember it was the first of many times I allowed myself to cry with a patient’s family and share a powerful human moment while at work.

As I reflect on this poignant memory which in many ways influenced my decision to become an Infectious Diseases physician later in life, I also recognize how incredibly lucky I am to have made it into adulthood to be in the position to make that decision. You see, as someone born and raised in sub – Saharan Africa, the part of the world with the highest rates of infant mortality, surviving childhood is not to be taken for granted. It implies surviving several bouts of malaria and other preventable infectious disease conditions which claim the lives of hundreds of children daily in this part of the world. In 2017, the risk of a child dying before their first year was highest in the WHO Africa region (51 per 1000 life births), over 6 times higher than in the WHO European region (8 per 1000 life births). With these staggering figures, effective interventions to target infant mortality in the region can’t come quickly enough and for the first time in many years this summer holds a special hope.
Bioko and Malawi – A time of hope and promise

In April of this year, the announcement of the upcoming malaria trial set to begin in 2020 in Bioko, Equatorial Guinea raised a lot of hope in the infectious disease community for several reasons. This is not the first malaria vaccine trial in the region but for the very first time we have a malaria vaccine which going into a large clinical trial, has proven to be up to 100% efficacious in laboratory studies and in small numbers of healthy volunteers. Previous malaria vaccine trials have had mixed degrees of success, with some showing only partial efficacy and presenting serious challenges for effective widespread delivery.
For example, the WHO also in April of this year, announced its approval of widespread testing for GlaxoSmithKline’s Malaria vaccine Mosquirix® in 360,000 children in Malawi and two other African countries. GSK’s vaccine in smaller pilot studies, has only shown about 30-40% efficacy in preventing severe malaria and about 60% reduction in severe anemia – one of the major complications of the disease. It requires four injections before the age of two which presents a unique challenge in synchronizing delivery with national immunization schedules. Furthermore, it is unclear how long-lasting these limited levels of protection provided by the vaccine will be. These issues have raised skepticism on the WHO’s decision to move forward with widespread testing of this vaccine with many holding the view that resources would be better served towards expanding prevention strategies with better established track records.
The fact that the WHO will move forward with its development and distribution highlights the urgency with which effective solutions to fight malaria and other causes of infant mortality are needed. Malaria still causes 450,000 deaths every year with most of these being children under the age of five in sub-Saharan Africa and even a 30-40% reduction in severe disease is worth a shot. The WHO hopes that the expanded trials of the vaccine will provide information on its real-life efficacy and guidance on making recommendations for its broader use across the region, results that we all eagerly await.
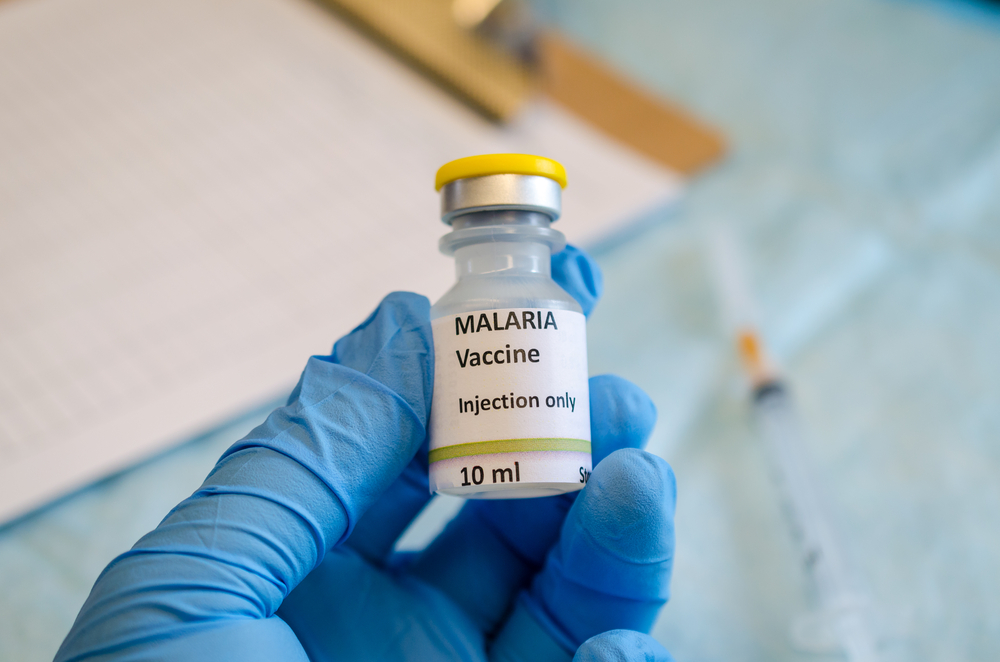
The vaccine, named PfSPZ which will be tested in Bioko – Equatorial Guinea, was developed by Sanaria® a biotech company based in Maryland. It is slightly different from other malaria vaccine candidates which have used protein components of the parasite to generate an immune response. PfSPZ uses weakened whole malaria parasites, that do not cause infection but are able to generate a robust protective immune response against live malaria infection. In this initial phase of large-scale testing, 2100 people between the ages of 2-50 years will receive the vaccine providing information on its safety and efficacy in a malaria endemic region.
The small study which demonstrated the efficacy of the PfSPZ vaccine enrolled 31 healthy adults ages 18-45. Participants received three intravenous doses of the vaccine at 8 weeks intervals and 19 weeks after the final vaccine dose were exposed to the same strain of malaria used to manufacture the vaccine and the vaccine was found to proffer high levels of protection lasting up to eight months.

The ultimate test for this vaccine will be how it performs in an area of high malaria endemicity against different strains of the parasite. Another challenge is the requirement for intravenous administration for up to three doses. Most childhood vaccines are easily administered by oral, intradermal and intramuscular routes and a vaccine requiring the additional skill of intravenous administration adds a layer of complexity to large scale distribution in resource limited settings. With that being said the future for malaria prevention in sub-Saharan Africa and the implications this will have for infant mortality in the region is promising with much to look forward to going into 2020. The only way to find a refine solutions that work will be through the information obtained from these essential trials.
Azithromycin for everyone?

Before I got the chance to fully digest my excitement over the upcoming Malaria trials, more news of another potentially cheap and simple intervention to reduce childhood mortality in sub-Saharan Africa landed earlier this month. Researchers from University of California San Francisco published the findings of their trial providing the longer-term assessment of the use of Azithromycin in reducing childhood mortality in Africa.
These results were a follow-up on an initial study published 10 years ago which incidentally found that administering a single dose of Azithromycin to children aged 1 to 9 years as a measure to prevent Trachoma, resulted in a 50 percent decrease in childhood mortality in children who received the intervention. Trachoma is the leading cause of preventable blindness in the world and Azithromycin is active against the infection that can lead to irreversible damage to the eyes. This initial trial was carried out in Ethiopia, a country with an estimated infant mortality of 58 per 1000 live births and which also has a high prevalence of Trachoma. Being able to reduce infant mortality by such significant numbers through a simple intervention set the stage to test the intervention on larger numbers of children in the region.

With funding secured through the Gates foundation the intervention was expanded to 200,000 children in Niger, Tanzania and Malawi with slightly less impressive reductions in childhood mortality of 14-18% noted. How the intervention works to reduce mortality is not completely understood but the researchers propose that the antibiotic likely provides some protection against common respiratory and gastro-intestinal pathogens which cause disease in young children in developing countries. Legitimate concerns over the risk of increasing antibiotic resistance overtime if this intervention were to be used more broadly cast a shadow over these encouraging results. The fear was that widespread distribution of an antibiotic will select for drug resistant pathogens and limit effective treatment options for serious infections in the future.

The latest studies published in the New England Journal of Medicine by the same research group allay some of these fears but also confirm some of the concerns previously mentioned. They have been able to show at the three-year follow-up mark in Niger, that childhood mortality in children receiving Azithromycin twice a year remains the same, suggesting that the intervention does not lose its efficacy overtime. Concomitantly a separate sub-study by the same group also noted an increase prevalence of pneumococcus (a bacteria which causes pneumonia and sinus infections), resistant to antibiotics in the same class as the intervention drug Azithromycin. Whether this raised prevalence of drug resistant Pneumococcus should completely nullify the clear benefits of the intervention is not an easy question to answer.
Childhood Mortality in the Developing World – No easy solutions!

When it comes to reducing childhood mortality in developing countries, there are no simple or straight forward answers but there is certainly promise and reason for optimism especially at this time. It is unlikely that we will find a perfect solution which does not present any challenge be it in terms of delivery, level of efficacy or perceived adverse secondary effects. What I do know is that at intervention even with only a 10% mortality benefit would have given Amina a better shot at making it to adulthood an achieving her full potential and for that reason I have hope in what the future holds.
Written by: Boghuma K Titanji






